|
Tuesday, November 27, 2007
>hello, hello, HELLO everyone!
It’s finally the time to say goodbye! (please don’t miss me, *hands tissues to you*) sadly we’ve reached the end of our wonderful fermentation journey! (*cue; awww..) this was a highly enriching and informative journey, which brought us a lot of fun and new insights to the wired glass bottle (fermentor)! –beams-

From the fermenter experimental journeys, we’ve learnt like 1276727367 things!
Now identifying the different fermenter parts is chicken feet! Preparing the equipment prior to use and media for inoculation is no sweat too! Scaling up from shake flasks to the bioreactor and inoculating the fermenter is easy peasy! Obtaining a sample during the fermenter run and monitoring the culture hourly is like “No problem man!” Harvesting cells, isolating desired product and purifying it is like eating with our eyes closed!
No sweat at all!
How we love hands on experiences! Hahahaha.. so many great benefits! (eg: embedding hard core information into our wooden heads. haha…)
Oh well, but all good things always come to an end somehow. we will definitely be missing the refreshing experiments and the impressive equipment used in the practicals (although it was all hard work). the experiments were all extremely enjoyable and it was quite a blast learning as a team (Group 5A! yay!).
 It was nice while it lasted, oh well! –shrugs- aahaha…like I said before, all good things always come to an end one day and today is the day. hope you guys love reading about us... thanks loads for dropping by and visit!
♥ karen & lucas.
Monday, November 26, 2007
> And below is the table for the absorbance reading of the fractions that contain the GFP.
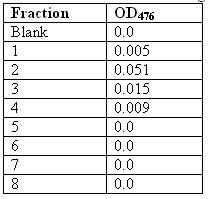 and this is the graph. (: and this is the graph. (:
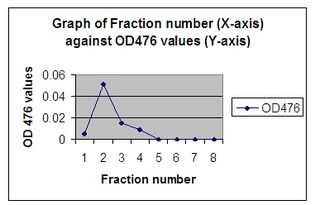 The principle of this chromatogram is that the more green fluorescent protein (GFP) that is eluted in the tube, the more fluorescence it will give out; therefore, the more light will be absorbed and the higher will the absorbance reading be.
For the graph above, the absorbance value of the blank was not included because it would only destroy the structure of the graph. Instead, it was just used as a negative control to compare the results with. Interpreting from the graph above, fraction 1 had an absorbance of almost 0 and therefore had almost no GFP in it. (=\) This is because the GFP will take some time to flow through the column and be eluted out. (hais)
Fraction number 1 would most probably contain the ammonium bicarbonate buffer which is used to rinse the column. On the other hand, fraction 2 had the highest peak in the graph – highest OD reading, showing that it had the most GFP in the tube. This is where most of the GFP has reached the bottom of the column and was eluted out. The value of OD reading started to decrease for fraction number 3 and subsequently fraction 4. This is the elution of the left over GFP. There was nil absorbance value for fraction 5 to 8 showing that all the GFP has already been eluted out. We can conclude that there was GFP collected in tube 2 to 4 only. -shrugs-
The protein with a MR of 50, 000 kD would definitely elute in a fraction before the GFP which has a MR of 27, 00 kD only. This is because the heavier molecule would definitely reach the bottom of the column first because it would not interact much with the column due to its larger size. (Fat!)
This is the principle of size exclusion chromatography where the smaller molecules would spend more time interacting with the column. Therefore, in this case, the GFP is the smaller molecule and would spend more time interacting with the column which slower down the flow rate and thus, would reach the bottom later than the protein with MR of 50, 000. -beams-
Don't you just LOVE us? Hahahahahah...
♥ sajini & siti.
> For purification, extraction that was carried out during the isolation step will be purified using Gel Permeation or Size Exclusion Chromatography. this method uses a column of polymer resins (Sephadex G75).
 owww... is this protein tech or bioprocess tech?? owww... is this protein tech or bioprocess tech??
the resins contains teeny weeny pores in which the molecules are small enough to diffuse into. (woohoo..nice and slim!) The larger molecules would flow through the column faster when the extract is added into the column. (Fat lah!) The smaller molecules on the other hand will spend more time interacting and diffusing into the pores of the gel resins. Hence separation of different molecules by size is achieved! (*clap clap*) First up, we had to label eight test tubes (1 to 8) and a blank where the blank was filled with 2 ml of ammonium bicarbonate. Next is to carefully drain the column into a waste beaker until the buffer is just even with the top of the gel bed, ensuring that the column does not run dry. (water water!) we had to use a disposable plastic pipette to transfer the cell-free extract to the top of the gel by gently swirling the pipette around the inside edge of the column, just above the top of the packed matrix. (Mmmm!) 
WATCH OUT! dont even think about disturbing the matrix.
next in line, we had to collect the eluant in each test tube filled to the 2ml mark. taking of fractions begin now. The stopcock is opened slowly and the sample is allowed to flow completely into the gel bed and 50 mM ammonium bicarbonate buffer is added continuously to the top of the column while the fractions are taken. (Must not to disturb the gel bed ok! maintained 2-3 cm "column" of buffer on the top of the gel column to provide consistent flow of buffer through the chromatography matrix alright?)
 zhi kang. dont hide la. get ready the tubes.
after collecting 8 tubes of 2ml fractions, then we wash the column with buffer till about 60 ml is collected in the beaker. and watching it drip drip drip... makes us sleepy...zzzz
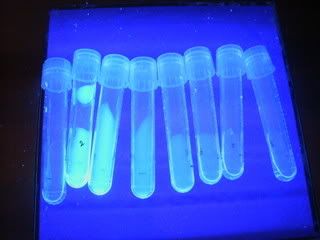 tadah~! the 8 tubes~! with different amount of GFP concentration
♥ sajini & siti.
> after harvesting, the product was sent for isolation and purification.
for isolation, first we collect 10 ml of culture broth into a test tube. after that we have to centrifuge at 100,000 rpm for 5 minutes. (this is basically to separate the cells from the culture broth. cells are denser so a pellet is formed at the bottom.) now it's time for us to view the product in the pellet under the UV light. (sun-tan lotion anyone?)
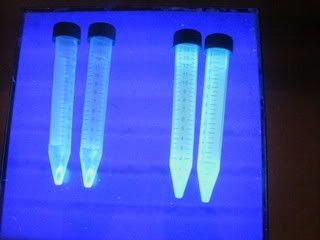 Ahh~!~! ET phone home... Ahh~!~! ET phone home... GFP is an intracellular product, therefore the bacterial cells need to be lysed first to release the protein. well, the 3 methods used in carrying out cell disruption on the bacteria cells are as follows...(*sing* I will follow you, follow wherever you may go...)
Method 1: Using enzymes.
okay. first we have to resuspend the pellet in 500 µl of TE buffer (pH 7.5) using a micropipette until there are no clumps. eventually bubbles were observed due to forceful suspension. poor babies. ):
 okay. Sajini... you smell nice whenever you go. (:
next we had to add 2 drops of lysozyme to the resuspended cell pellet. (initiate enzymatic digestion of the bacteria cell wall. now it's time to allow the enzymes to act for 15 minutes.)
Method 2: Freezing and Thawing
the best part of the entire experiment is when we placed the tube in liquid nitrogen until the contents froze. not only had the contents frozen, many other items froze too..
 okay. settle down. no murmuring...
next in line was the thawing of our frozen babies in warm water. we had to repeat the cycle 2 more times. (this is to complete the rupturing of the bacteria cell wall)
 hmm. this is FUN. Baby~ Baby~ one more time~!
freezing and thawing add mechanical stress to the cell wall as the cell water content expands (when frozen) and contracts (when thawed).
Method 3: Sonication
cell disruption is completed by the process of sonication where ultrasonic waves cause the bacteria cell wall to implode under the vibrational pressure. the brave one steps in the sonication lab to conduct the sonication process.
 wait a minute, it's not the 'brave one', but instead the 'brave two'.
 DJ King Kang Kong in the house.
the sonication process is performed in ice to prevent the DENATURATION of the product as a lot of heat will be produced 4 cycles of 25 seconds with 10 seconds rest in between sonication cycles is carried out. err its just so boring waiting for the entire 4 cycles to be over.
next in line is to centrifuge for 20 minutes at 10,000 rpm after cell disruption. oh no!!! both the centrifuge machines are in use. now how... OCM to the rescue...
 is a bird? is a plane? no, it's OCM to the rescue!!
next we had to resuspend the pellet using 400 µl of TE buffer. and now its time for us to view the product under UV light (again).
 it's glowing! it's glowing!!
so we NOW know that the supernatant contains the product! BINGO!!!
♥ sajini & siti.
>whee! day 5 of experiment, last day!!
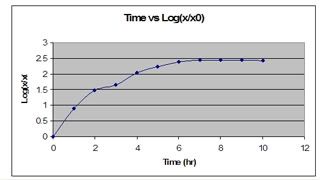
these are the absorbance value of the 10 samples at OD600, taken on day 3.
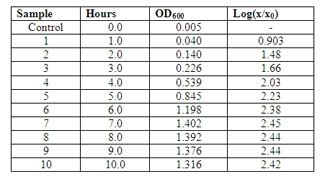 and the graph. (:
spectrophotometer is the instrument for measuring and comparing the intensities of common spectral lines in the spectra of two different sources of light. for UV/VIS measurements, a spectrometer can be configured with a range from 200 to 1100 nm and a spectral resolution of 1.4 nm FWHM.
further a combined deuterium-halogen light source is needed. 600nm was used to provide higher resolution measurements. as no dilution was done, the cells might be too concentrated (too many babies~!) thus, lead to higher amount of absorbance. if absorbance value is more than 1, it will not be too accurate. technically speaking, dealing with a smaller absorbance help increase the sensitivity of the measurement. (recalling from the 4 concepts of analysis)
♥ flavia & faith.
Thursday, November 22, 2007
> btw. pH, temperature and dissolved oxygen are 3 very important control philosophy.
the temperature used to cultivate E.coli must be regulated. at a high temperature, they will be kill off as their enzymes are denatured (this will be a waste of our efforts). if they are growing at a much slower rate, the operating cost in cultivating E. coli will increase.
E. coli prefers to grow at a pH range of 4 to 8. as they are living organism, they produce wastes and require oxygen. the wastes that these organisms produced are very acidic (ouch~! i doubt ours are the same.) if the solution is not neutralize, or maintain at the optimum pH, the environment will not be conducive for their survival. if the pH is kept under the optimum pH over a period, they can only survive for a few hours to a day.
dissolved oxygen is crucial for their survival. without oxygen, they will not be able to carry out their biological processes and multiply in numbers. if so, due to the insufficient oxygen, they will die (and yes, just like any other living organism!!!). without having enough cells to replace the dead cells, soon, there will be no more live cells.
♥ flavia & faith.
> this is what we did on day 4.
we harvested 10ml of the fermentation broth from a sterile, disposable tube and placed under UV light to check for presence of desired protein produced by the cells, after which we were all shipped to see the history plot and managed to derive some conclusions.
GFP is a secondary metabolite. the product was harvested at the late exponential phase. this is due to the expression of the machinery for secondary metabolite biosynthesis occurs only in late exponential growth stage. however, theoretically secondary metabolites are harvested in the stationary phase-idiophase (just like their puberty stage).
below shows the history plot of the process.
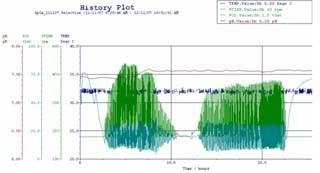 the controlling parameters for the history plot are: pH temperature dissolved oxygen
the pH has to be maintained at pH 7.5, since the pH of an actively growing culture will not remain constant for long. pH measurement was done via pH probe which is connected to a pH meter. when the pH of media drop below 7.5, the media becomes acidic, and the computer control system sends signals to pump in sodium hydroxide into fermentor, if pH of media rises above 7.5, causing the medium to be alkaline, sulphuric acid is pumped.
temperature has great influence in the biological processes in a cell through kinetics effects (rate of reaction) and catalytic effects (enzymatic activity). a temperature probe is used to indicate if there are any changes in the temperature of the medium. at 32 oC, desired product GFP folds and fluorescence optimally. any higher temp would denature the protein, the rate of flow of cooling water or steam through the jackets ensures that the temp of fermentor is constant.
E.coli being a living organism, it requires ample amount of air to survive, or the cells will die. hence its an aerobic fermentation. therefore dissolved oxygen here is crucial. the partial pressure of O2 is set to be 20% like in the open environment. when pO2 is not 20%, the rate of injection of air bubbles by the sparger is adjusted.
♥ flavia & faith.
Wednesday, November 21, 2007
> day 3 of our experiment!! (:
let's recap on what we have done for the last 2 days:
seed culture from the streak plate was transferred to the shake flask --> magnified from the shake flask (E.coli cells were PROLIFERATING!!)
dont you wanna know what we did to them?!
okay. we inoculated 100ml of seed culture from the shake flask to the fermentor, this is in line with increasing the yield of desired protein (the GFP), in other words, we were gradually scaling up the fermentation process. how interesting!
steak plate --> shake flask --> fermentor
of course, the parameter has to be set first:
pH 7.5 stirred speed (min 10%, max 90%, control to AUTO) pO2 set point (set point 20%, control to AUTO. stir to CASC & AIRFLOW to CASC) airflow (min 25%, max 100%)
with the computer control system, the conditions of the fermentor at different time of the day are collected and stored also known as data logging. the system also helps perform calculations based on the mathematical models of the process also known as data analysis. the system also process control and optimization. it sends signals to pumps, valves, switches and alarms to adjust the conditions when no one is there to manually check. to sum up, IT JUST MAKE LIFE EASIER FOR US~! (how great with machines arnd??) oh yes, and we rmbed to take out 10ml medium before inoculation which is for our _____. (in case if u didnt know we meant BLANK!)
samples were drawn to monitor cell growth and product formation hourly, and we divided ourselves into pairs. (how wise were we??! (: )
each pair assigned to each hour to take the sample, at the same time computer data logging was set up by our lab tech so we could monitor the fermentor parameters.
to be continued...
♥ flavia & faith.
Tuesday, November 20, 2007
> day 2... continues...ahem.. according to the prac manual, we're suppose to set up the bioreactor as well. but.. it's so nice of the lab tech to set it up for us. think maybe they were afraid that we wld mess up the whole bioreactor? anyway. this is what we HAVE to do if we were to set up the bioreactor by ourselves. first, we must calibrate the pH electrode by using the standard buffer solution, meaning we must measure the optimun pH levels with 2 buffers, a pH7.0 buffer and either an acid or a base buffer, depending on the range of pH that is to be measured.
then have to install the pH, pO2, foam and level probe...
the height of the foam and level probe must be adjusted if necessary. After connecting the 4 probes, more connections.. the weak acid, base and antifoam..
other accessories (air inket, exhaust filter, cooling jacket sampling unit etc) for the 'pretty' bioreactor must also be connected before using it. After dressing up, the bioreactor is ready for sterilization. Those equipments that are heat sensitive are being wrapped up in aluminium foil..
the pO2 probe must be polarized then calibrate after sterilizing. polarization means putting the probe in an O2 environment to make sure it adapt to the O2 in the bioreactor. this will ensure that the probe will read and measure the amount of O2 at a high rate when the probe is polar. and the last connection is made with the peristaltic pumps, a positive displacement pump used for pumping fluids contained within a flexible tube fitted inside a circular pump casing..
♥ zhao zhi & zhi kang.
> today, we're able to see the E.coli colonies growing on the media. (woweee!)
Look at my BEAUTIFUL plate...
want to see more??

check out my website at mocca.com NOW!! (just kidding)
we then transferred several colonies on the streak plate into the shake flask containing 100ml LB media then put it in the oven (incubator i mean.) at 32oC for 24hrs. the rest 1900ml of LB broth, tgt with the fermenter, is sent for AUTOCLAVE! ampicillin is added after autoclaving to prevent it from denaturing and rendering it useless, due to the high heat. the ampillicin used to stop other bacteria from grwoing inside the medium. it also kills off E.coli which do not carry the GFP protein ad they are not ampicillin-resistant, resulting in only the GFP-carrying E.coli multiplying. 
mr. fermenter in aluminium spacesuit.
♥ zhao zhi & zhi kang.
Monday, November 19, 2007
> okay. here we are again. (:
after learning abt the fermenter, we went on to learn how to make the media for the seed culture and prepare the bioreator for the scaling up process.
to prepare 2 litres of the LB medium, we dissolved 50g of LB powder in 2 litres of distilled water.

the kitchen is over there... (always look at the direction)did we mention abt wat is the recipe of the LB powder?
it consists of: bacto-tryptone yeast extract NaCl distilled water pH of 7.5
bacto-tryptone provides peptides, peptones and essential amino acids for bacteria growth, and the yeast extract provides organic compounds that are required for bacteria propagation. the NaCl provides ions necessary for transport and osmoic balance in the media.
as we are adding the LB powder, magnetic stirrer is used to dissolve the powder at a faster rate.
prepare to be hypnotized...after finished preparing the media, we incolated pGLO transformed E.coli onto a LB/Amp/Ara plate and incubated for 24hrs, meaning we have to come back tmr. ):
the ababinose in the plate induces the production of the GFP. and sterile techniques are used in seed preparation. gloves were worn during the transfer of the bacterial colonies from the plate into the shake flask. the entire procedure was also done in a laminar flow cabinet to prevent contamination from the environment. the media in the flask also contained small amounts of ampicillin which would remove any contaminants that were to entire the media ( the bacteria used is ampicillin resistant).
btw, the purpose of this inoculation is to scale up. step-wise scale up is done to allow the cells to reduce the lag phase. if the cells were transferred for example from the agar plate as individual colonies directly into the bench top fermentor, they would have a very long lag phase and the whole process of scaling up and production is slowed down dramatically. a lot of energy is being wasted as well
the reason being that if the lag phase is long, then the overall fermentation time would be longer and thus the fermentor would have to run much longer to obtain the same amount of products. instead, if the cells were scaled up using the step-wise method, it would have a shorter lag phase and thus a shorter overall fermentation run.
♥ zhao zhi & zhi kang.
> okay. first up was to familiarize with the parts and components of microbial and mammalian bioreactors, and to know the basis operation procedure of the bioreactor.
The different parts of the bioreactor are identified and labelled. 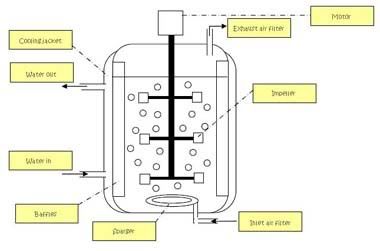
The following table shows the functions each parts of the bioreactor.
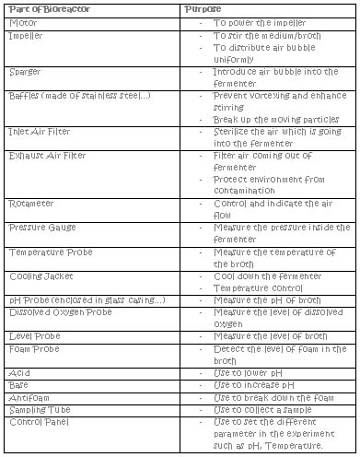
after that, we realised that the mammalian cell bioreactor is actually slightly different frm the microbial bioreactor that we are using. the mammalian cell bioreactor uses modified impeller (marine propeller type) and modified sparger for gentle aeration, whereas the microbial bioreactor uses flat-blade propeller. the mammalian cell bioreactor does not have the baffles which reduces the shear force that is crucial for the mammalian cells as they are very delicate.
that's all for now. shall post up some more info abt wat we did for the preparation of that stinky LB broth and streaking of the agar plates. (:
♥ delia & alvin.
> our first day for bioprocess tech practical. whee!!
our grp started off with getting to know the different parts and components of the bioreactor, and the functions of the different parts, while the other grp started off with the preparation of the stinky LB broth.
being brief-ed on the workflow of the prac. and this is wat we're gonna do for the prac.
familiarization with the bioreactor and its operation
- we would be learning abt the different parts and components of the bioreactor, and their functions as well. we wld also be learning how to operate the bioreactor. equipment, media and seed culture preparation
- the media and seed culture will be prepared to get ready for the scale-up fermentation. the bioreactor is also being set-up. inoculation, fermentation and monitoring
- scale-up fermentation is being carried out to increase the yield of the desired product. the appropriate control parameters for the bioreactor is being set. manual sampling is taken to monitor cell growth and the formation of the product. isolation and purification of product
- the product is collected and isolated by 3 methods: using enzymes, freezing and thawing and sonication. purification is then done to collect the purified GFP
♥ delia & alvin.
|